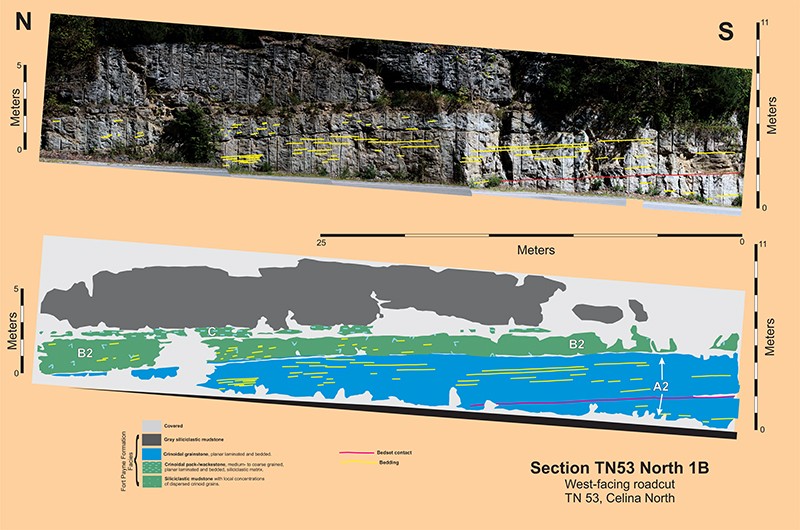
Sections TN53 North
TN 53 Roadcut north of Celina, TN
(4.75 Km north of Obey River bridge)
Section TN53 North 1A
Section TN53 North 1A describes a heterolithic channel-fill some 100-meters long and 4-5-meters thick; this channel-fill has a minimum aspect ratio of 22:1. The fill is at least 80% siliciclastic mudstone with dispersed crinoid grains; however, there is enough crinoid grainstone present to define the steeply inclined southern margin and portions of the basal erosion surfaces. Five different stratigraphic units comprise this channel-fill: A1, A2 (the thickest and most extensive crinoid grainstone), B1, B2 (the major siliciclastic mudstone fill), and B3; see Section TN53 North 1A. Crinoid packstone unit C is a sheet-like deposit that disconformably overlies this channel-fill.
The crinoid grainstone comprising unit A2 defines the steeply inclined southern margin of this channel-fill and, furthermore is the most readily apparent stratigraphic body exposed at this roadcut. The planar internal bedding is parallel the steeply inclined southern portion and downlaps onto the nearly horizontal northward portion of its basal erosion surface. The channel in which A2 was deposited had a minimum depth of 4-meters. However, beneath the A2 directly overlying the flatter, nearly horizontal northern portion of its basal erosion surface, there is yet another crinoid grainstone, the less than 30 cm-thick unit A1. This oldest part of this channel-fill sequence has a basal erosion surface and is capped by a 10- or so centimeter thick siliciclastic mudstone which is overlain by the basal erosion surface of unit A2. It is reasonable to suppose that the steeply inclined A2 erosion surface truncates the original, southern part of the A1 basal erosion surface. There is no direct evidence supporting this supposition, however, neither is there any evidence supporting the continuation of the A1 surface below the A2 and out into the adjacent siliciclastic mudstone. The upper contact of Unit A2 appears to be an erosion surface, based on local relief. A 10-cm thick layer of siliciclastic mudstone occurs on top of this surface. This mudstone is truncated by the basal erosion surface of overlying unit B1. In summary, the steeply inclined southern margin of the channel-fill sequence is defined by a crinoid grainstone A2, which is bounded on bottom and top by erosion surfaces. Unit A1, a decimeter-thick crinoid grainstone, occurs beneath the northern part of A2. Both units A1 and A2 are capped with centimeter-thick layers of siliciclastic mudstone.

The difficulty with determining the actual cross-cutting relationships of the basal erosion surfaces of units A1, A2, and B1 is illustrated in Section TN53 North 1A-Detail 1. The A1 basal erosion surface disappears to the south into a covered interval and to the north into siliciclastic mudstone. The A2 basal erosion surface also disappears to the south into a covered interval and to the north into a covered interval and/or siliciclastic mudstone. The erosion surface at the top of the A2 crinoid grainstone likewise disappears to the north into a covered interval; it probably does truncate the basal erosion surface of the A2 grainstone, however, this geometry cannot be verified. The B1 basal erosion surface disappears to the south into a covered interval and becomes somewhat intermittent to the north because of local disappearance into siliciclastic mudstone. The simplest interpretation of these ambiguous geometries is that the A2 basal erosion surface is the major bounding surface of this channel-fill sequence and that the A1 deposit is a minor, pre-existing channel-fill that has been subsequently truncated.
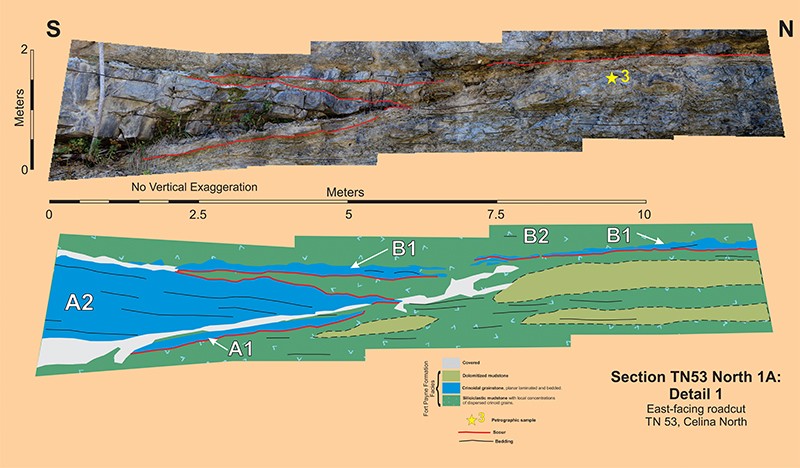
Channel-fill sequence B is the predominant unit exposed in Section TN53 North 1A and consists of three separate units: a) the decimeter-thick, basal crinoid grainstone B1 which occurs immediately above an erosion surface, b) the 4-5 meter-thick, middle siliciclastic mudstone B2 which comprises the vast majority of sequence B, and c) the up to 2 meter-thick, upper crinoid packstone B3 which is organized into gently inclined beds. These B3 crinoid packstones have an internal structure similar to the classic Bouma turbidite, see Section TN53 North 1A-sample 1.

Section TN53 North 1B is located on the opposite side of TN53 some 20-meters to the east. The channel-fill consists of the following: a much thicker unit A2, a thinner unit B2, and a unit C of essentially the same thickness; the contacts between these units are covered. Planar bedding characterizes the two bedsets of Unit A2. Measured perpendicular to bedding, unit A2 in section 1B is much thicker than its counterpart in section 1A, 4.5 meters minimum versus 2.5 meters maximum. This difference suggests that unit A2 could have been deposited in a body similar to a fluvial point bar that was thinning as it curved to the north.